The First Clinically Validated Breathing Device
Designed for Postpartum Recovery
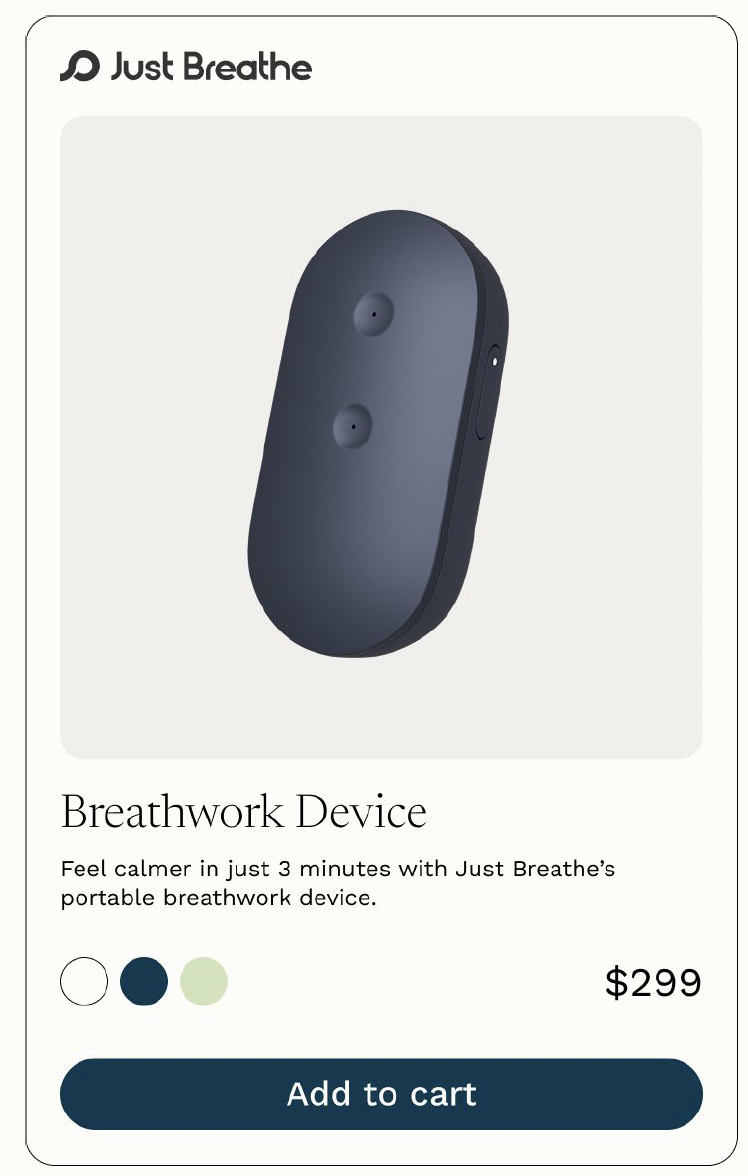
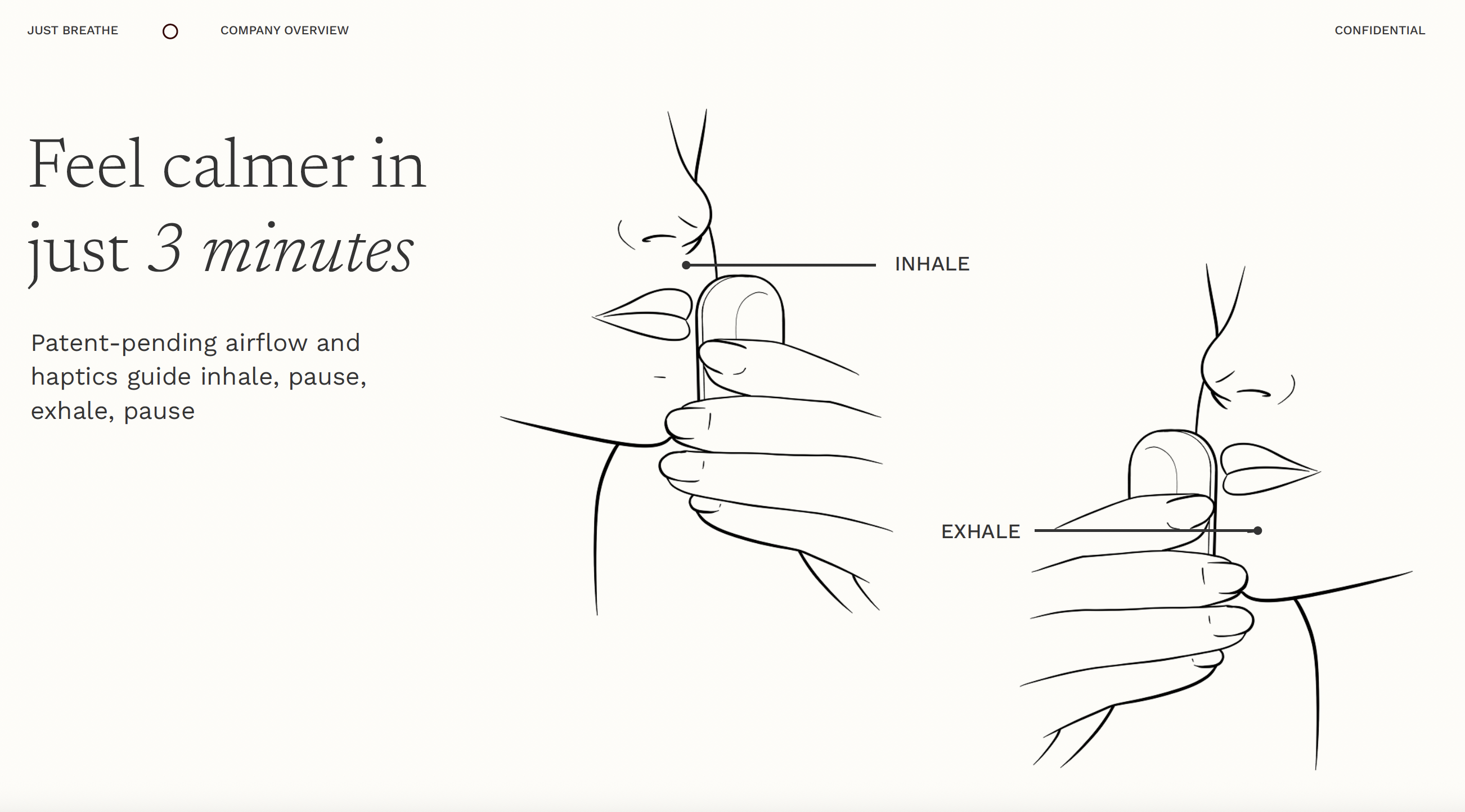
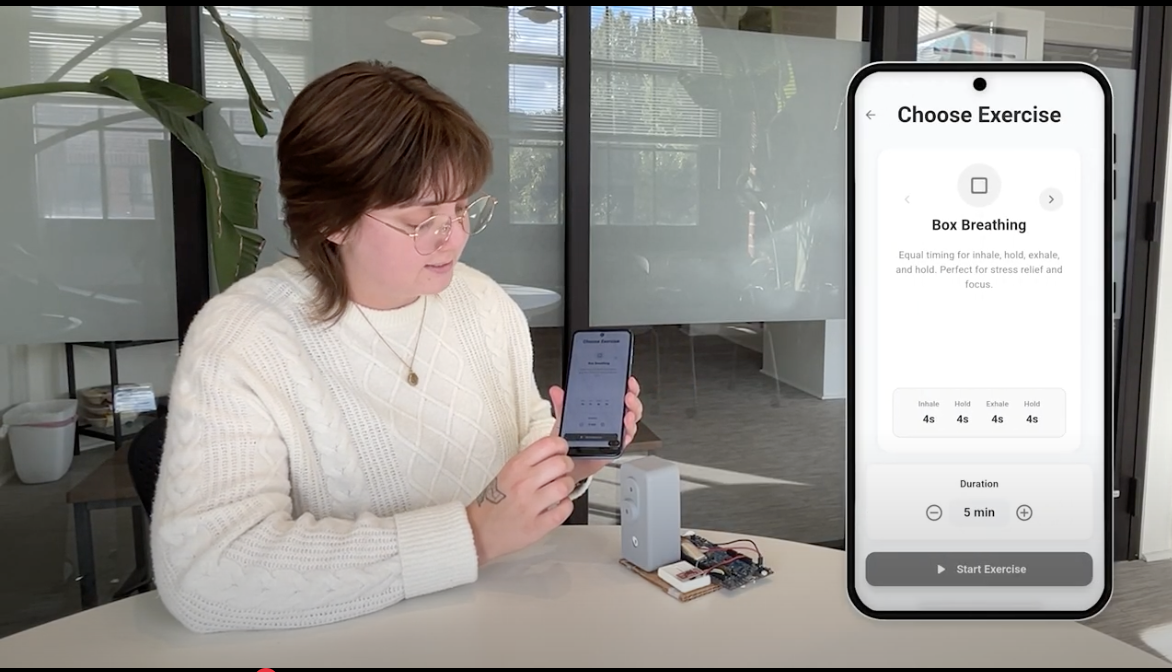
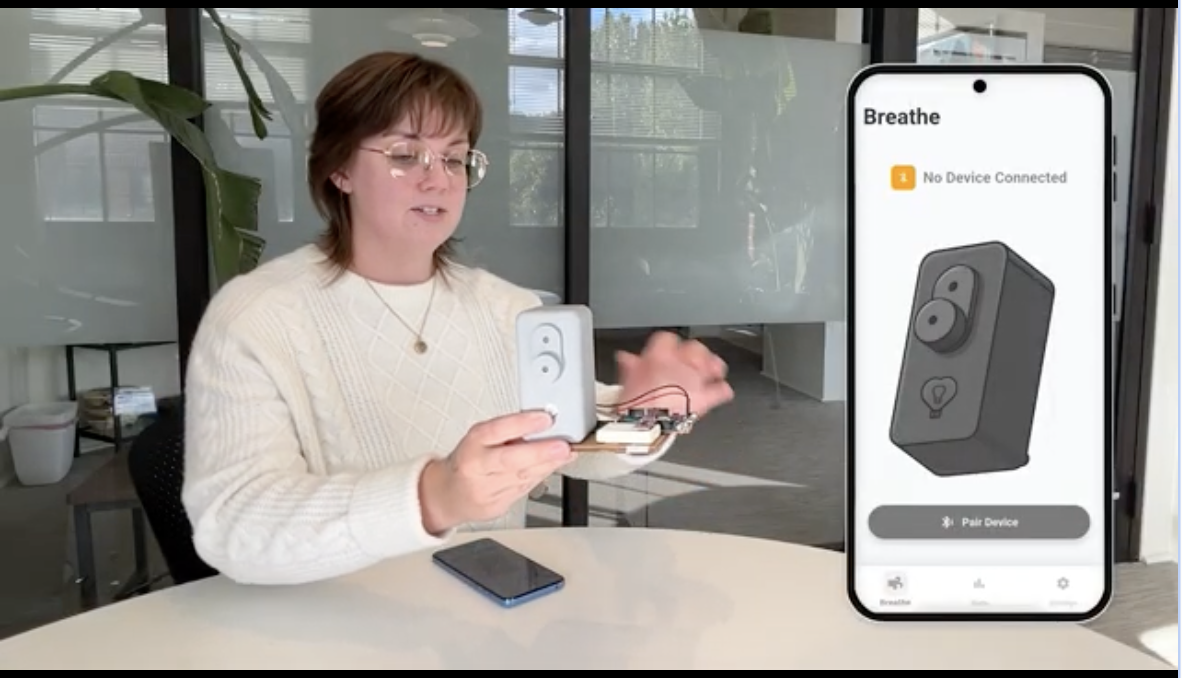
Just Breathe combines airflow-guided breathing with real-time biofeedback to help patients reduce anxiety, improve resilience, and feel supported - starting with the 700,000+ women affected by postpartum mood and anxiety disorders each year.
Backed by a peer-reviewed publication and clinical study conducted with Brown University and Women & Infants Hospital
Be first in line when we launch. No spam, ever.